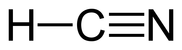Hydrocyanic acid is mainly produced by direct reaction of alkanes (organic moelcule known as hydrocarbons) with ammonia, used as a byproduct of the manufacturing of acrylonitrile (chemical compound used in the production of plastics and acrylic fibers). Its molecular formula is similar to HCN.
Hydrocyanic acid can be produced by reacting ammonia, methane and oxygen over a platinum-rhodium cataylst at 1000 deg C. Hydrogen cyanide was used a as chemical warfare weapon to cause disturbance, diesease or death during war.
Hydrocyanic acid is too deadly to be used directly in any commercial products.
Hydrocyanic acid can be produced by reacting ammonia, methane and oxygen over a platinum-rhodium cataylst at 1000 deg C. Hydrogen cyanide was used a as chemical warfare weapon to cause disturbance, diesease or death during war.
Hydrocyanic acid is too deadly to be used directly in any commercial products.
Acrylonitrile and Hydrogen Cyanide Structure